Chemistry laboratory
Chemistry laboratory
Traditional wet-chemical processes are executed in the chemistry laboratory within research or third-party projects. Additionally, samples are prepared for subsequent equipment-based measurements and preliminary investigation. The laboratory operates besides photometer and automatic titration units several other pieces of equipment.
Rapid chloride permeability test (RCPT)
This method accelerates the migration of chloride ions into a sample by applying an electric current externally. The procedure permits the evaluation of the penetraton resistance against chloride of concrete made with different types of binders. This is an important information for reinforced concrete structures close to streets where deicing salt is used.

Figure: RCPT-cell
Thermographical detection of dissolvers
The ingress of dissolvers into constructions materials represents a serious thread for health, environment and fire safety. While slowly evaporating liquids can be detected rather easily visually on the split surface of a sample tested, this is not possible for rapidly evaporating liquids. The latter leave for a certain time span (up to 15 minutes) a “cold trace” caused by the evaporation which can be visualized and subsequently measured by using a thermographic camera.
Figure: Thermographic evaluation of a concrete sample, which was penetrated by hexane up to a depth of 7 cm
X-ray diffraction
X-ray diffraction
The X-ray diffractometer is used for the investigation of crystalline mineral phases present in construction materials such as cement, concrete, sand-lime bricks and many more. Dry powders and tempered samples can be studied in a temperature range from 3 °C to 70 °C. Transient or so-called in-situ measurements observe the alteration of the mineral phase content in the course of a reaction in a sample as for instance in hydrating cement.

Figure: PANalytical Empyrean X-ray platform with copper anode und PiXcel1D Detector
Due to their small wavelength of approximately 10-10 m (1 Å) X-rays are applied for measurements in the atomic dimension in order to determine characteristic periodic structure with dimensions also ranging in the Å-scale. The method is based on the diffraction of monochromatic X-rays on the planes of crystals. The diffracted waves exhibit a path difference and an interference described by Bragg’s law. The incident angle θ is related directly to the spacing d of the lattice planes and thus to the crystalline structure of the mineral phase under investigation. The information obtained permit the quantitative and qualitative analysis of crystalline mineral content of a sample. Rietveld’s method enables the quantitative analysis by fitting a diffractogram based on existing structural data such that it coincides with the measured diffractogram.
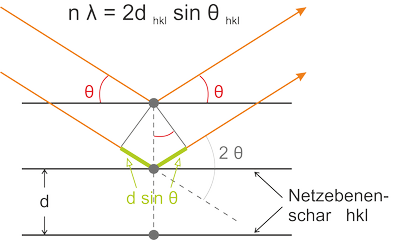
Figure: Bragg's law – Condition for constructive interference
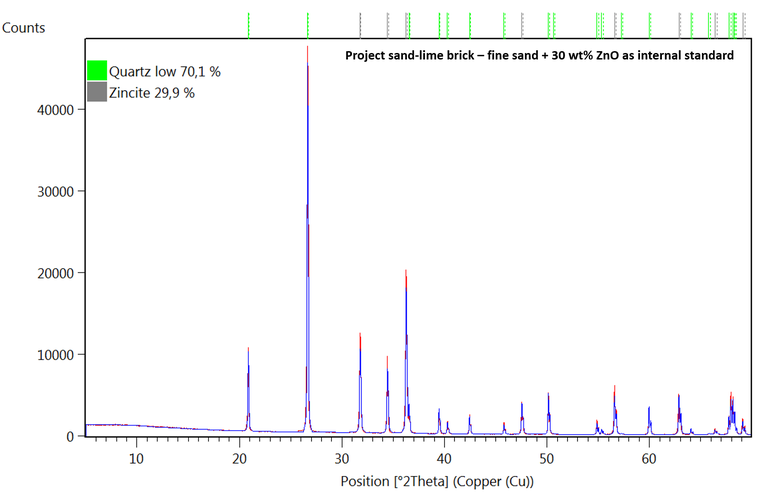
Figure: diffractogram of fine sand
Differential thermal analysis
Differential thermal analysis
Thermogravimetric analysis and differential thermal analysis (TGA-DTA)
While a substance is heated slowly on a sample holder its changes in weight and enthalpy (heat absorption or release) are registered. The method is also referred to as simultaneous thermal analysis (STA) and can be applied to any material undergoing a reaction in the temperature range from 20 to 1600 °C.
Observing the substances which are either released during the temperature rise (e.g. water of crystallization) or are absorbed (e.g. oxygen) as well as emerging structural modifications yield an insight into the reaction mechanisms and permit the quantitative determination of the permuted substances. Main fields of application are the investigation of mineral materials (e.g dehydroxilation of clays or formation of clinker phases in cement raw meal) and the characterization of plastics. Complex mixed substances are difficult to interpret since several processes may superimpose each other.

Figure: Simultaneous thermal analysis - Netzsch STA 449 F3 Jupiter
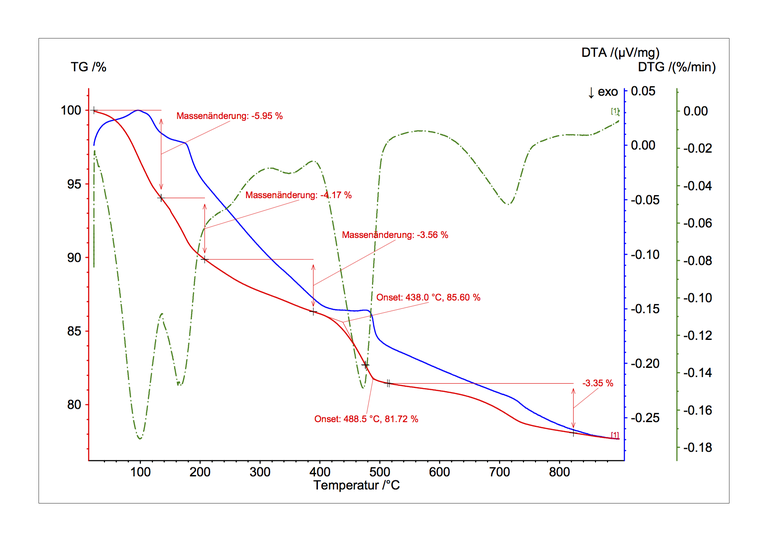 Figure: Temperature-dependent release of water permits the investigation of the cement hydration
Figure: Temperature-dependent release of water permits the investigation of the cement hydration
TGA-IR-coupling
Simultaneous thermal analysis (STA) does not always provide clear conclusions about which substance is causing a specific change in weight and enthalpy. This holds especially in case of complex mixed substances. Here, an analysis of the gases released during the STA-measurement is necessary and can be accomplished by coupling the STA with the FT-IR-spectrometer.

Figure: Nicolet iS10 FT-IR spectrometer coupled with TGA-IR gas cell coupled with transfer line to the STA 449 F3 Jupiter
The coupled system is suitable for the identification of carbon carriers (e.g. petroleum) in mineral construction materials and for analyzing polymers.
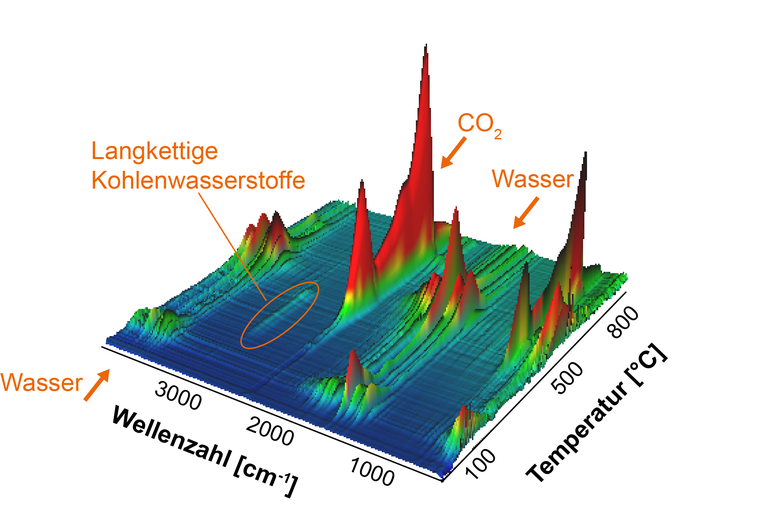
Figure: Thermal analysis coupled with FT-IR-spectroscopy
Infrared spectroscopy
Infrared spectroscopy
Fourier-transform-infrared spectrometer (FT-IR spectrometer)
IR spectroscopy is a physical analysis method which uses infrared radiation and belongs to the molecular spectroscopy methods. Materials absorb certain frequencies when exposed to infrared radiation. The absorption bands are related to molecular energy states. IR spectrum can be visualized in a graph of infrared light absorbance vs. frequency or wavelength. The position of the absorption bands is characteristic for particular chemical bonds (⇒ functional groups) which facilitates a structural determination (⇒ identification of materials). A limitation of the method is given since the interaction between electromagnetic radiation and a molecule occurs for IR-active molecules only. During measurements the wavelength is between 7800 cm-1 and 350 cm-1 and diamond or Germanium are used as ATR (attenuated total reflection) crystal plates.

Figure: Thermofisher Scientific Nicolet iS10 FT-IR spectrometer
Chemical analysis
The following measurements are possible:
- Measurement on liquid and solid samples (⇒ studying of the surface)
- Identification of organic substances (⇒ structural determination) and aging of plastics
- Quantitative identification of known substances
- In-situ-spectroscopy (⇒ series spectrogram)
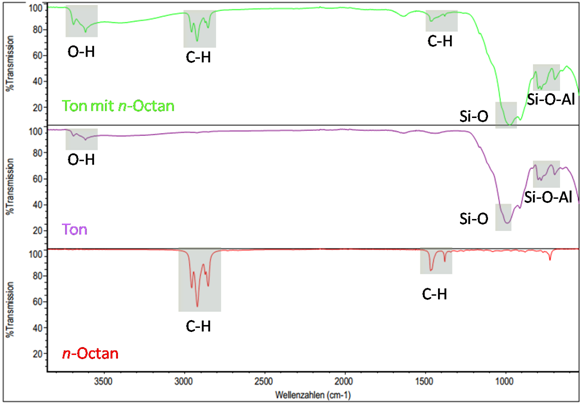
Figure: Identification of organic substances in a raw clay via IR
Gas analysis
An additional feature is the gas analysis. The gas mixture is fed into the IR- gas cell of the IR-spectrometer during measurement. An example of such transmission measurement is the coupling with the simultaneous thermal analysis (STA).
