Abstract
Oxidation flow reactors (OFR) were demonstrated to fill the gap between oxidation states observed in ambient aerosols and the ones which could be reached in smog chamber experiments. Up to now, several OFRs and operation modes have been proposed to simulate atmospheric chemistry in the laboratory, e.g. to study yields of secondary organic aerosol (SOA) from precursors (Peng & Jimenez, 2020), but also for toxicological exposures (Offer et al., 2022). However, high exposures of OH radicals, high UV light intensity and short OFR residence times has been criticized and proposed to generate aged aerosol being different in chemical composition compared to ambient aerosols.
Peng & Jimenez (2020) developed a model to evaluate the comparability to atmospheric aging conditions involving ratios of atmospheric oxidants OH, O3, O(1D) and O(3P) as well as photolysis for classification of “good”, “transient” and “risky” conditions. However, Peng & Jimenez (2020) state that OFR-generated SOA may differ in properties compared to ambient aerosols.
Experimental and Results
We generated SOA from toluene (tol-SOA) in der OFR “Photochemcial Aging Emission flowtube Reactor” (PEAR) at “good”, “transient” and “risky” conditions (Fig.1 ) as well as in the ILMARI smog chamber at the same OH exposure of 0.9 to 1.2∙1011 s cm-3. The gas phase and particularly the SOA was investigated concerning its molecular composition by a set of state-of-the-art analytical techniques.
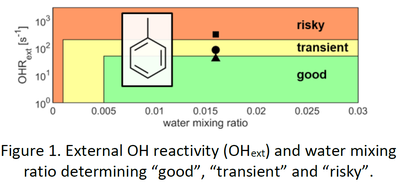
Tol-SOAs have similar properties in bulk composition, becoming slightly less oxidized from “good” to risky” conditions, but reveal clear differences in molecular composition (Fig. 2). Furthermore the size mode of SOA particles increased from below 20 nm (a/b) over 55 nm (c) to 200 nm derived from SMPS.
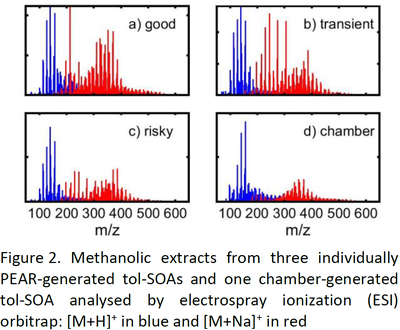
Surprisingly, the chemical composition and properties of chamber-generated tol-SOA were most similar to the one from “risky” OFR conditions. Hence, our study challenges the current concept for assessing OHR chemistry only from gas phase reaction and calls for further research, aiming to harmonize OFR conditions and chemistry in the atmosphere.
This work was supported by the Helmholtz International Laboratory aeroHEALTH (InterLabs-0005).
Ihalainen, M., Tiitta, P., Czech, H., Yli-Pirilä, P., …, Sippula, O. (2019) Aerosol Sci. Technol., 53 (3), 276-294.
Offer, S., Hartner, E., Di Bucchianico, S., …, Zimmermann, R. (2022) Environ. Health Perspect., 130 (2), 27003.
Peng, Z., and Jimenez, J. L. (2020) Chem. Soc. Rev., 49 (9), 2570–2616.